100% Genuine
Sealed pack
Pharmacist-verified
Quality assured
Pan-India delivery
Standard shipping

100% Genuine
Sealed pack
Pharmacist-verified
Quality assured
Pan-India delivery
Standard shipping
We'll let you know the moment SAFLUTAN-S EYE DROPS is back in stock.
Important advisories specific to this medicine.
Pregnancy
Use with caution / Consult doctor (use only if benefit outweighs risk)
Alcohol
No known interaction
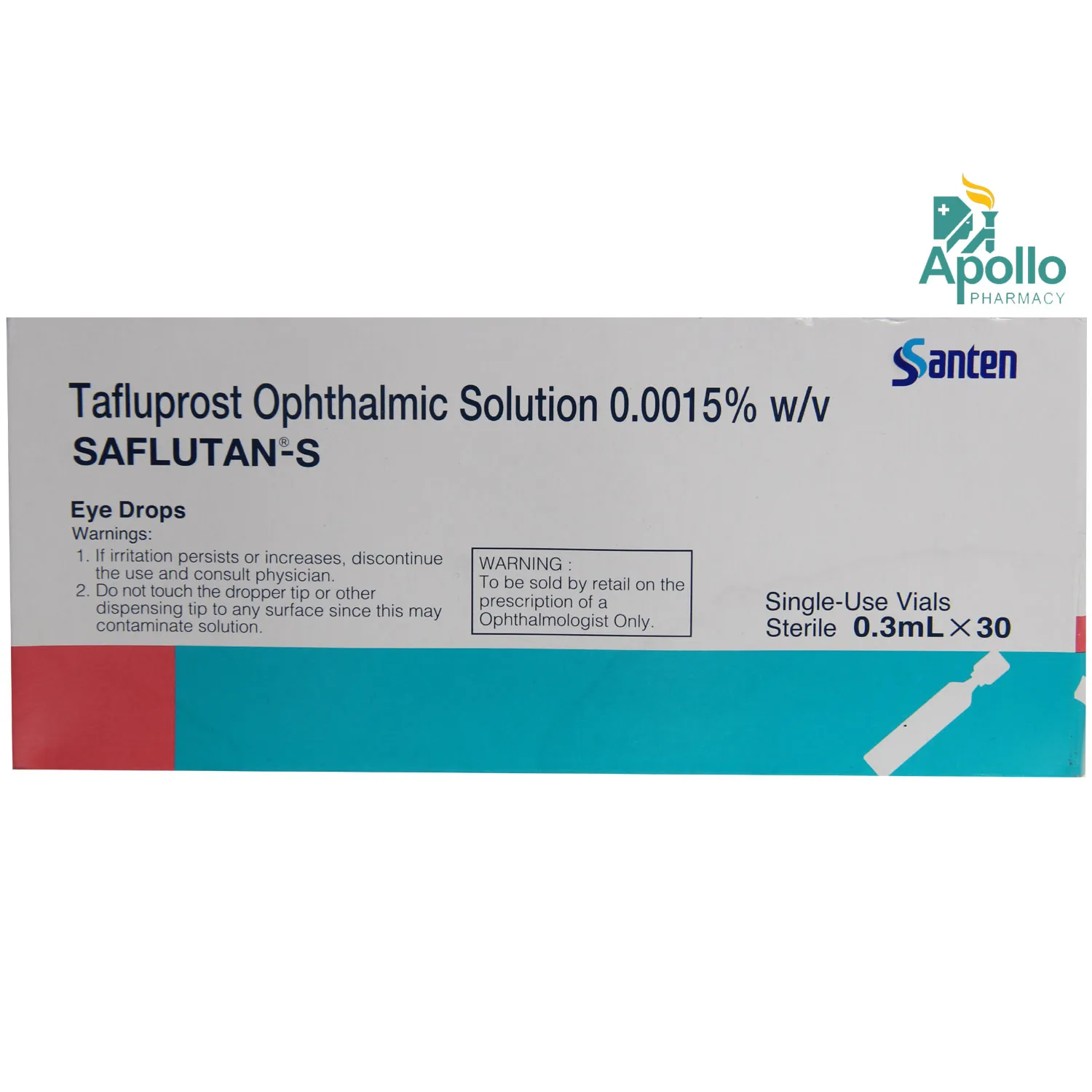
Reduction of elevated intraocular pressure (IOP) in open-angle glaucoma and ocular hypertension when combination therapy is required.
Eye irritation/burning, conjunctival hyperemia (redness), itchy eyes, dry eyes, blurred vision, increased eyelash growth, darkening of iris/eyelid skin, headache, eyelid irritation
Tafluprost is a prostaglandin F2α analogue that increases uveoscleral outflow of aqueous humour, lowering IOP. Timolol is a non-selective beta-blocker that reduces aqueous humour production, further decreasing IOP.
Brand
SANTEN LIMITED
View all products from this brand
Every order from PingMeDoc Pharmacy is reviewed by a licensed pharmacist before dispatch. Prescription medicines ship only after the Rx is verified.
Manufacturer: SANTEN LIMITED
Medical disclaimer
Product information is for awareness only and does not replace medical advice. For Rx medicines, a valid prescription is required. Consult your doctor or pharmacist for personalised guidance.
Sources: Manufacturer labeling/SmPC, established medical literature on tafluprost and timolol, ophthalmology treatment guidelines
Distributor inventory | Drop
Reduction of elevated intraocular pressure (IOP) in open-angle glaucoma and ocular hypertension when combination therapy is required.
Tafluprost is a prostaglandin F2α analogue that increases uveoscleral outflow of aqueous humour, lowering IOP. Timolol is a non-selective beta-blocker that reduces aqueous humour production, further decreasing IOP.
For ophthalmic use only. Instill 1 drop in the affected eye(s) once daily, preferably in the evening, or as prescribed by the ophthalmologist. If using other eye drops, keep a gap of 5–10 minutes; remove contact lenses before use and reinsert after 15 minutes (if applicable).
Eye irritation/burning, conjunctival hyperemia (redness), itchy eyes, dry eyes, blurred vision, increased eyelash growth, darkening of iris/eyelid skin, headache, eyelid irritation
Use with caution in patients with asthma/COPD, bradycardia, heart block, or heart failure due to timolol systemic absorption; may cause bronchospasm or slow heart rate. Prostaglandin analogues may cause permanent iris pigmentation changes and eyelash changes. Avoid touching dropper/vial tip to prevent contamination. Use caution in history of uveitis, macular edema, or aphakia/pseudophakia with torn posterior lens capsule. Do not stop abruptly without medical advice; monitor IOP regularly.
Other beta-blockers (oral or ophthalmic) may increase risk of bradycardia/hypotension; calcium channel blockers (verapamil/diltiazem), digoxin, antiarrhythmics (e.g., amiodarone) may enhance cardiac effects; clonidine withdrawal may worsen hypertension; CYP2D6 inhibitors (e.g., fluoxetine, paroxetine, quinidine) may increase timolol levels; other IOP-lowering eye drops may have additive effects.
Store as per pack label; typically protect from light and moisture. Keep in original pack, do not freeze. Single-use vial: use immediately after opening and discard any remaining solution.
Taflotan-T Eye Drops, Tapcom Eye Drops, Tafluprost/Timolol Eye Drops (generic), Xalacom Eye Drops (latanoprost+timolol), Ganfort Eye Drops (bimatoprost+timolol)
For ophthalmic use only. Instill 1 drop in the affected eye(s) once daily, preferably in the evening, or as prescribed by the ophthalmologist. If using other eye drops, keep a gap of 5–10 minutes; remove contact lenses before use and reinsert after 15 minutes (if applicable).
Store as per pack label; typically protect from light and moisture. Keep in original pack, do not freeze. Single-use vial: use immediately after opening and discard any remaining solution.
Yes, this product requires a valid prescription (Rx). Upload it during checkout when prompted.
Yes. Prices and availability can change based on inventory and serviceability. The latest details are shown at checkout.
Select a variant, add to cart, enter delivery address, upload prescription if required, and pay securely.
Other products patients also browse in this category.
Same brand, other products you might also want.